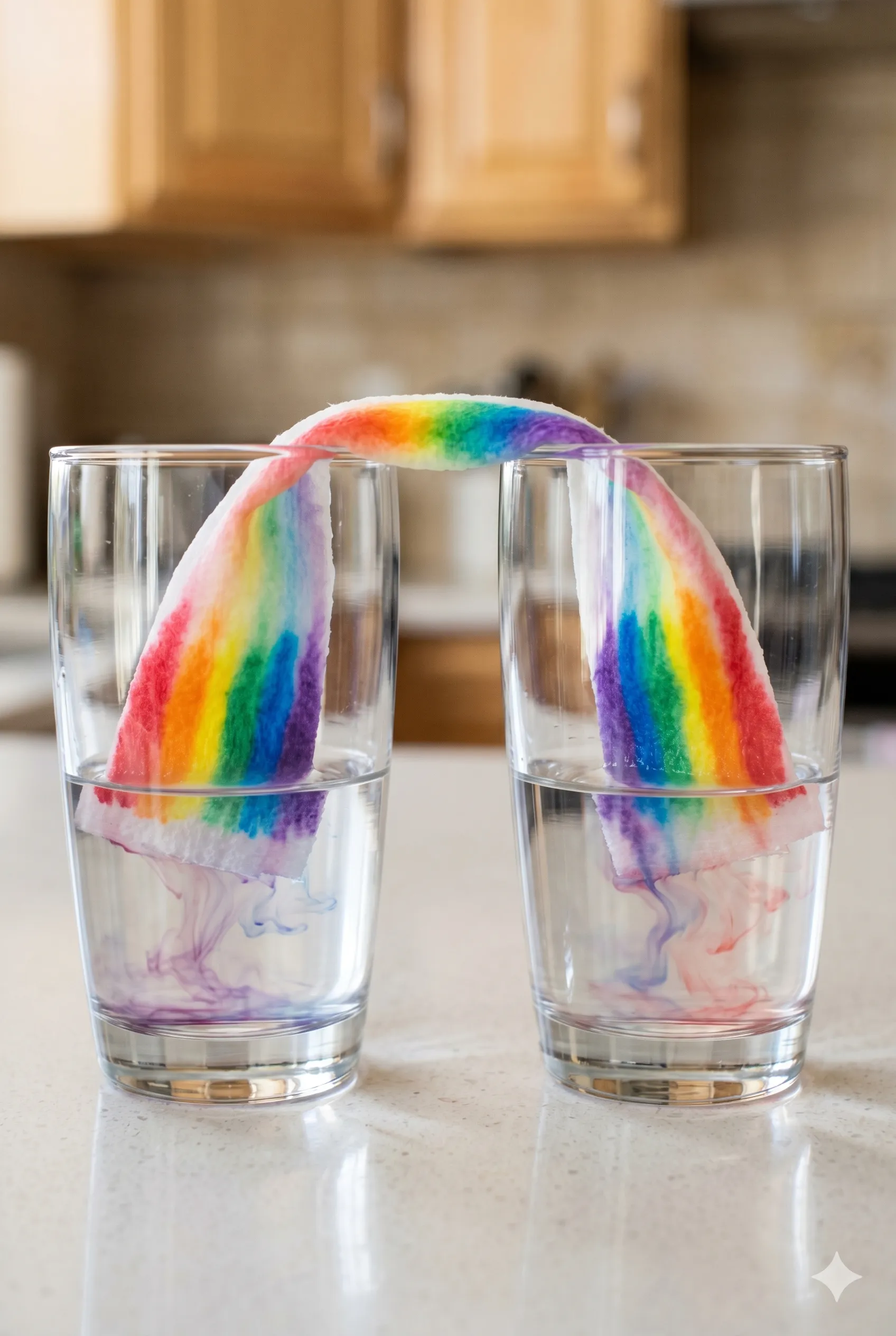Some science experiments deliver a result. This one delivers a revelation. The rainbow paper towel experiment sits in a rare category of activities where the finished product is genuinely beautiful, the process is completely absorbing, and the science underneath it is rich enough to fuel a real conversation about chemistry, color, and the physical properties of water.
And yet, most setups get it at least partially wrong. Either the colors bleed into a muddy brown mess, the paper towel tears mid-experiment, or the “rainbow” ends up looking more like a beige smudge with optimistic labeling. Almost every one of those failures traces back to a small handful of avoidable mistakes — in the materials chosen, the markers used, or the way the paper towel is handled.
This guide covers everything: the real science, the precise setup, the most common mistakes, and several variations that turn a single activity into a full afternoon of guided discovery.
This short video shows the rainbow paper towel experiment for kids:
Here’s the Real Reason Colors Travel on Paper Towels
Most people assume this experiment is purely about capillary action — water climbing through the paper towel fibers and dragging color along with it. Capillary action is definitely part of the story, but the more interesting science happening here is something called chromatography.
Chromatography is a technique used in real chemistry labs around the world to separate mixtures into their individual components. The word comes from the Greek chroma, meaning color, and graphein, meaning to write — literally “color writing.” In its simplest form, it works by exploiting the fact that different molecules travel at different speeds through a material when carried by a solvent.
In this experiment, the paper towel is the stationary phase — the material the molecules travel through. The water is the mobile phase — the solvent doing the carrying. The ink molecules in your markers are the mixture being separated. Different ink pigments have different molecular weights and different levels of attraction to the paper fibers. Heavier molecules, or ones more strongly attracted to the paper, travel more slowly. Lighter or less attracted molecules travel faster.
This is why a single black marker dot, when water travels through it, often separates into surprising rings of purple, blue, red, and yellow — colors that were always in the ink but invisible because they were blended together. The paper towel and the water between them act as a molecular sorting machine.
Capillary action is the force doing the actual carrying — water climbing through the paper towel fibers by adhesion (attraction to the fiber surfaces) and cohesion (water molecules pulling each other along). Understanding both processes together gives kids a genuinely complete picture of what they’re watching.
What You’ll Need
One of the best things about this experiment is how little it costs and how accessible the materials are. However, a few specific choices make a significant difference in the quality of results.
- Paper towels — thick, high-quality kitchen paper towels work best. The thicker the towel, the more defined the color separation. Avoid thin, single-ply towels as they tear easily when wet and don’t produce clean chromatography bands.
- Washable markers — this is critical. Washable markers contain water-soluble dyes that separate beautifully when water travels through them. Permanent markers use alcohol-based or oil-based inks that water cannot dissolve or carry, meaning no separation will occur. Use a full set of colors including black, which produces the most surprising and dramatic separation results.
- Water — plain tap water works perfectly here, unlike the instant ice experiment. Room temperature is ideal.
- Small cups or bowls — one per paper towel strip if doing the standing version of the experiment
- A ruler and scissors for cutting consistent strips
- A pencil for marking dot placement
- Optional: A spray bottle for a controlled misting method, and clothespins or tape for hanging finished strips to dry
Don’t Ignore Your Marker Choice — It Changes Everything
This deserves its own section because it is the single most common reason this experiment disappoints. If you pick up whatever markers are closest and they happen to be permanent markers, the experiment will produce almost no visible separation. The ink simply won’t dissolve in water.
Washable markers (Crayola washable markers are a widely available and reliable choice) contain dyes that dissolve readily in water and separate into distinct bands as they travel through the paper towel. The black washable marker is particularly worth trying — most kids are genuinely surprised to discover that black ink is actually a mixture of multiple colors.
Gel pens can work to a limited extent but are less reliable than markers. Watercolor paint can produce beautiful results but behaves slightly differently, spreading more diffusely than a concentrated marker dot.
If you want to turn marker choice into part of the experiment, set up identical paper towel strips with the same color in both a washable and a permanent marker and compare results side by side. The contrast makes the point about water solubility absolutely concrete.
Step-by-Step: How to Do the Rainbow Paper Towel Experiment
Step 1: Cut your paper towels into strips approximately 3–4 centimeters wide and 20–25 centimeters long. Cutting along the perforated lines of the paper towel roll gives you a natural straight edge to work from. Consistent strip width matters because uneven strips absorb water at different rates across their width, which can skew the color bands.
Step 2: Use a pencil to draw a faint line across each strip about 3 centimeters from one end. This is your marker line — the point where you’ll apply the ink dots. Using a pencil rather than a pen is important here because pen ink would dissolve along with the marker ink and contaminate your results.
Step 3: Place marker dots along the pencil line. For the basic rainbow version, place one dot of each color — red, orange, yellow, green, blue, and purple — spaced roughly 1 centimeter apart across the width of the strip. For the chromatography investigation version, place a single large dot of just one color, particularly black, in the center of the strip.
Step 4: Pour a small amount of water into your cup — just enough to cover the bottom to a depth of about 1 centimeter. The water level is crucial: it must sit below your marker line. If the water touches the ink dots directly, the color will simply dissolve off rather than traveling upward through capillary action in a controlled way.
Step 5: Place the marked end of the paper towel strip into the water, making sure the water level stays below the marker dots. The bottom centimeter of the strip should be submerged. You can lean the strip against the side of the cup or hang the top over the cup’s edge. Some people find that folding the very top of the strip over the cup rim and securing it with a clothespin keeps the strip stable and prevents it from falling into the water.
Step 6: Watch and wait. Water will begin climbing the strip almost immediately. Within the first minute you’ll see the waterfront traveling upward. Within five to ten minutes, the colors will begin to move, separate, and in the case of black and brown markers, split into their component colors. Resist the urge to touch or move the strip during this phase — any disturbance can interrupt the separation bands.
Step 7: Remove the strip when the water front is about 2 centimeters from the top of the strip, or after approximately 15–20 minutes. If you leave it too long, the water reaches the top and starts running back down, blurring the color bands you worked to create. Lay the finished strip flat on a clean surface or hang it to dry.
Step 8: Once dry, compare your strips. Which colors traveled the furthest? Which stayed closest to the original dot? Did any colors separate into unexpected components? These are the questions that turn a craft activity into a science investigation.
The Cup-to-Cup Rainbow Version
This variation combines the rainbow paper towel experiment with the walking water experiment covered earlier, creating a visually spectacular result that works as a standalone display piece.
Set up seven cups in a row. Fill cups one, three, and five with a small amount of water and add red, yellow, and blue food coloring respectively. Leave cups two, four, and six empty. Fold paper towel strips lengthwise and bridge them between each pair of adjacent cups exactly as described in the walking water setup.
Now, before starting the water transfer, use washable markers to draw color bands directly onto each paper towel bridge in the dry section above the cups. As water walks across the bridges, it will carry and separate the marker ink simultaneously — producing both the walking water color-mixing effect in the cups and the chromatography separation effect on the paper towel bridges at the same time.
It takes longer to set up but produces one of the most impressive combined results of any household science experiment.
What Black Marker Really Looks Like
If you do nothing else from this guide, try this: take a single black washable marker and make one large, solid dot in the center of a paper towel strip. Set it up as described above and watch what happens.
Most kids expect the black to travel as black. What they see instead is the dot slowly separating — first into dark blue and purple bands near the bottom, then into reddish and yellowish rings higher up, sometimes with a distinct band of each color sitting at different heights along the strip. The black ink is revealed to be a layered mixture of multiple dyes that only looks black because they’re blended at full concentration.
This single demonstration is one of the most effective ways to introduce the concept that what we observe with our senses isn’t always the complete picture of what’s actually there — which is, in a way, one of the core lessons of science itself.
Fun Variations to Extend the Investigation
The Solvent Comparison
Run three identical strips with the same marker dot, but use different solvents: plain water, slightly salty water, and slightly soapy water. Do the colors travel at the same speed? Do they separate into the same bands? Salt and soap both change water’s surface tension and slightly affect how it interacts with both the paper fibers and the ink molecules, producing subtly different results.
The Paper Type Test
Cut strips from different absorbent materials — a coffee filter, a paper towel, a piece of newspaper, and a plain piece of printer paper. Use the same marker and the same water and compare how far the colors travel and how clearly they separate on each material. Coffee filters actually produce some of the cleanest chromatography results of any household material and are worth trying.
Reverse Chromatography
Instead of starting with a blended color and separating it, start with separate colors and try to predict what mixed color will form at the meeting point. Place a red dot on the left side of a strip and a blue dot on the right side, then dip both ends in water simultaneously. Where the two water fronts meet in the middle, the colors will mix — predicting the result before it happens turns this into a color theory and chemistry prediction exercise.
The Butterfly or Flower Art Project
Fold a paper towel in half and then in half again to create a small square. Apply marker dots in a pattern and dip one corner into water. The water spreads outward from the corner in all directions simultaneously, carrying the colors with it. When unfolded, the paper towel reveals a symmetrical, tie-dye-like pattern that looks nothing like what was expected. This version bridges science and art in a way that appeals to kids who might not immediately engage with the more structured investigation versions.
What Kids Learn From This Experiment
Chromatography is a real analytical chemistry technique used in forensic science to analyze evidence, in food science to test for artificial dyes, in environmental science to detect pollutants in water, and in pharmaceutical manufacturing to verify the purity of medicines. Kids who do this experiment at age seven are encountering the same fundamental principle that professional chemists use daily.
Mixtures and separation is a standard topic across elementary science curricula worldwide. Most textbooks describe separation techniques abstractly. This experiment makes the concept physical and visible in a way that no diagram can replicate.
Capillary action appears again here in a different context from the walking water experiment, reinforcing the concept through repetition in a new setting — which is exactly how scientific understanding deepens.
Color theory gets a chemistry dimension. Understanding that colors are physical mixtures of dye molecules, not just abstract categories, connects art class knowledge to science class knowledge in a way that feels genuinely illuminating.
Quick Troubleshooting Guide
| Problem | Most Likely Cause | Fix |
|---|---|---|
| Colors don’t separate at all | Permanent markers used instead of washable | Switch to washable water-based markers |
| Paper towel tears mid-experiment | Too thin or too wet too fast | Use thicker towels and lower the water level |
| Colors bleed into each other | Marker dots placed too close together | Space dots at least 1–1.5 cm apart |
| Water reaches top before colors separate | Strip too short or water level too high | Use longer strips and reduce water to 0.5 cm depth |
| Black marker stays black | Marker brand uses a single-dye black | Try a different brand of washable marker |
| Results look identical across all colors | Water is contaminated with previous color | Use fresh water for each new strip |
FAQ
Why does black marker separate into so many colors? Because most black marker ink is not made from a single black dye — it’s a blend of multiple colored dyes that together absorb most of the visible light spectrum, which our eyes interpret as black. When water carries those dye molecules through the paper towel at different speeds, the blend is physically pulled apart into its components.
Does the experiment work with food coloring instead of markers? Yes, with some differences. Liquid food coloring dropped directly onto a dry paper towel strip and then dipped in water will produce some separation, though the results tend to be less defined than with markers because food coloring is already in liquid form and spreads more before the water arrives. It’s worth trying as a comparison.
Can the finished strips be preserved? Yes. Once completely dry, the color bands are reasonably stable. Pressing the dry strip flat between two pieces of wax paper and laminating it, or simply displaying it flat under glass, preserves the result well. Some families frame particularly striking strips as art.
Why do some colors travel further than others? Because different dye molecules have different molecular weights and different levels of chemical attraction to the paper fibers. Lighter molecules with weaker attraction to the paper travel further before being slowed down. Heavier or more strongly attracted molecules lag behind. The distance a color travels is actually a measurable property of that dye molecule — in professional labs, this measurement is called the Rf value and is used to identify unknown substances.
Does water temperature affect the results? Slightly. Warmer water has lower surface tension and slightly higher molecular energy, which means it climbs the paper towel a little faster and may carry ink molecules slightly further before the strip dries out. For most home experiments the difference is minor, but testing warm versus cold water is a good variable to investigate if kids want to design their own follow-up experiment.
Conclusion
The rainbow paper towel experiment earns its popularity not just because the results are beautiful — though they genuinely are — but because the science underneath is real, significant, and connects to things that matter far beyond the kitchen table. Chromatography separates crime scene evidence. Capillary action moves water through every living plant on the planet. Color mixing is both an art principle and a physical chemistry process. Get the markers right, keep the water level below the ink dots, use a quality paper towel, and what you produce isn’t just a pretty strip of color. It’s a visible record of molecular behavior — which is, quietly, one of the most remarkable things you can make with a cup of water and a felt-tip pen.